
AI-Automated Detection of Hypertrophic Cardiomyopathy by Echocardiography: Training and External Validation (Late Breaking Science, ASE 2025)
Dr. Fawaz Alenzi, MD, MSc, FAHA, FACC, FASE is an Associate Professor of Medicine and Cardiologist at Duke Heart Center with a strong interest in multimodality cardiovascular imaging, including cardiac CT, cardiac MRI, and echocardiography. His research centers on driving innovation in cardiovascular imaging. At ASE 2025, Dr. Alenzi delivered a Late Breaking Science Presentation titled “AI-Automated Detection of Hypertrophic Cardiomyopathy by Echocardiography: Training and External Validation.”
This interview highlights the insights and reflections on this important study:
Can you tell us a little about your Late-Breaking Presentation: AI-Automated Detection of Hypertrophic Cardiomyopathy by Echocardiography: Training and External Validation, and what motivated this work?
This is one of the most promising studies we’ve worked on. Our motivation came from the fact that hypertrophic cardiomyopathy (HCM) is one of the most common inherited cardiovascular diseases and the leading cause of sudden cardiac death in young people. Yet, it remains significantly underdiagnosed. Research shows that nearly 80% of HCM cases go undetected. The reasons are multifactorial: the clinical presentation is often vague, and the manifestations can be very subtle.
What are some of the key challenges in diagnosing HCM?
Three main challenges come to mind:
- Subtlety. The manifestations are not unique. LV wall thickening may be borderline, and regional thickening can occur in areas that standard echocardiography or even an echocardiographer might miss.
- Variability. There is significant interobserver variability. Wall thickness measurements are not always reproducible.
- Access to expertise. Not every setting has access to expert cardiologists or advanced tools such as cardiac MRI, which are often needed for accurate diagnosis of HCM.
I understand you worked with the Us2.ai HCM model. Could you tell us more about how the AI model was developed and validated?
We were very fortunate to collaborate with Us2.ai. They developed a deep learning model for automated detection of HCM using routine echocardiographic video loops.
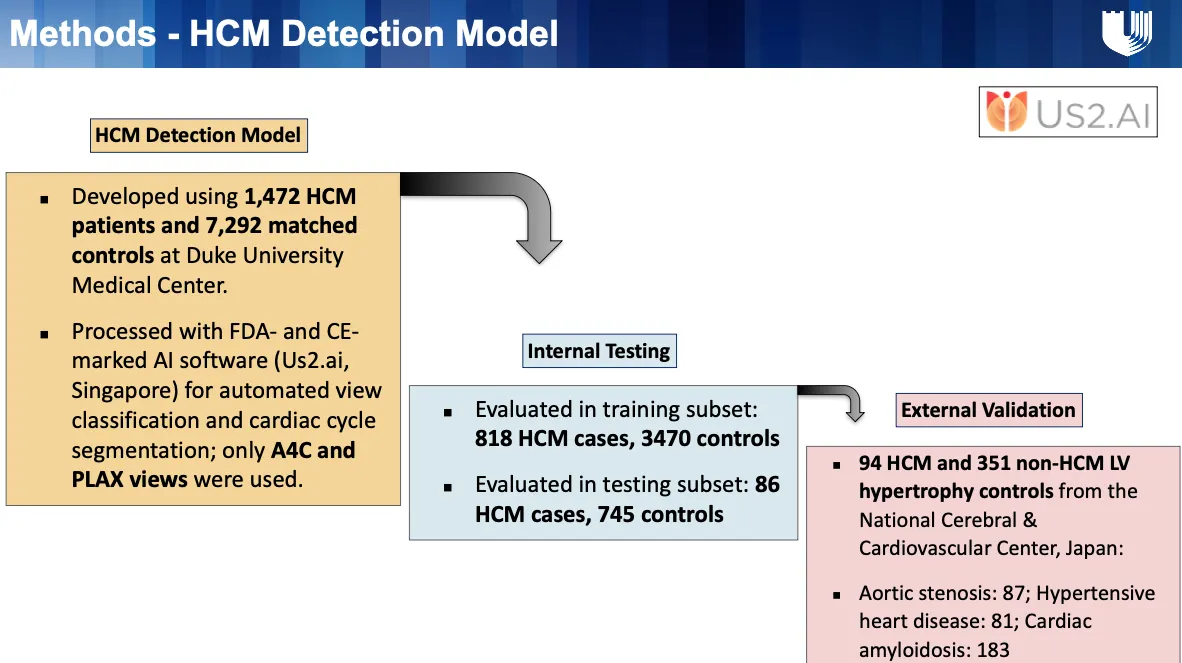
The training dataset included 1,472 patients with expert-adjudicated HCM and 7,292 controls without HCM. For internal validation, we held out 818 HCM cases and 3,470 controls from the Duke dataset. These were used to evaluate diagnostic accuracy.
To test generalizability, an external validation cohort was obtained from a Japanese multicenter registry. This cohort included 94 patients with clinically confirmed HCM and 351 patients with non-HCM LVH, consisting of 87 patients with aortic stenosis, 81 with hypertensive heart disease, and 183 with cardiac amyloidosis.
In addition to this, Us2.ai also developed a HOCM Gradients Model to measure LVOT gradients. This model was trained on 408 HCM patients from the Brigham Cardiovascular Imaging Core Laboratory. The pipeline incorporated automated Doppler view classification and Valsalva detection, focusing specifically on CW LVOT, CW Apical, and CW Mid-Ventricular views. From these, the model generated CW VTI segmentation masks to calculate peak gradients (Pmax). For external validation, AI-derived gradients were compared with ground-truth rest and provocation values from clinical reports of 712 HCM patients at Duke University Medical Center.

The performance results across both the detection and gradients models are very promising, and we are excited about the potential of these tools to enhance accuracy, efficiency, and scalability in HCM care.
Let’s dive deeper into your study. What results stood out to you most?
What excites me most is the consistency. The Us2.ai HCM detection model, validated internally on Duke data and externally on a Japanese multicenter cohort, achieved similarly high accuracy. This shows both reproducibility and reliability across diverse populations.
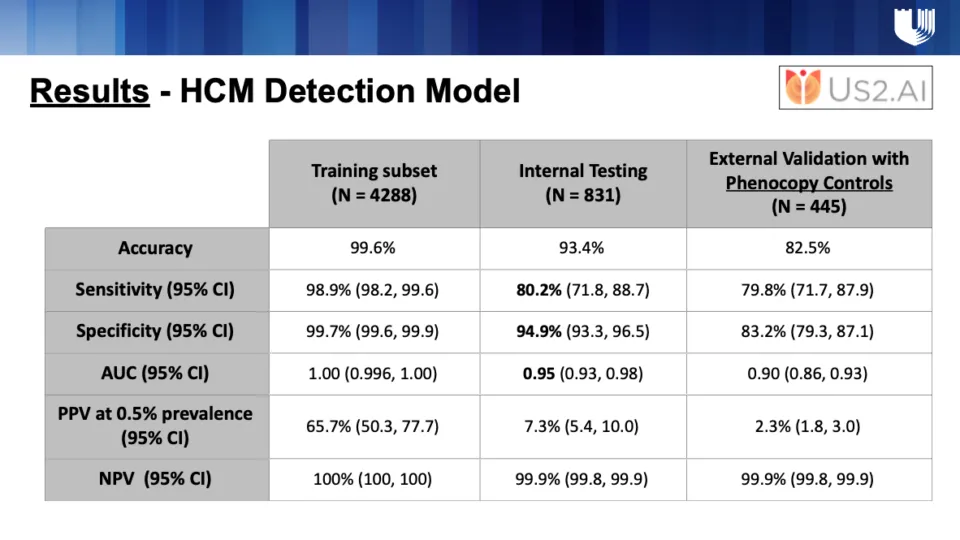
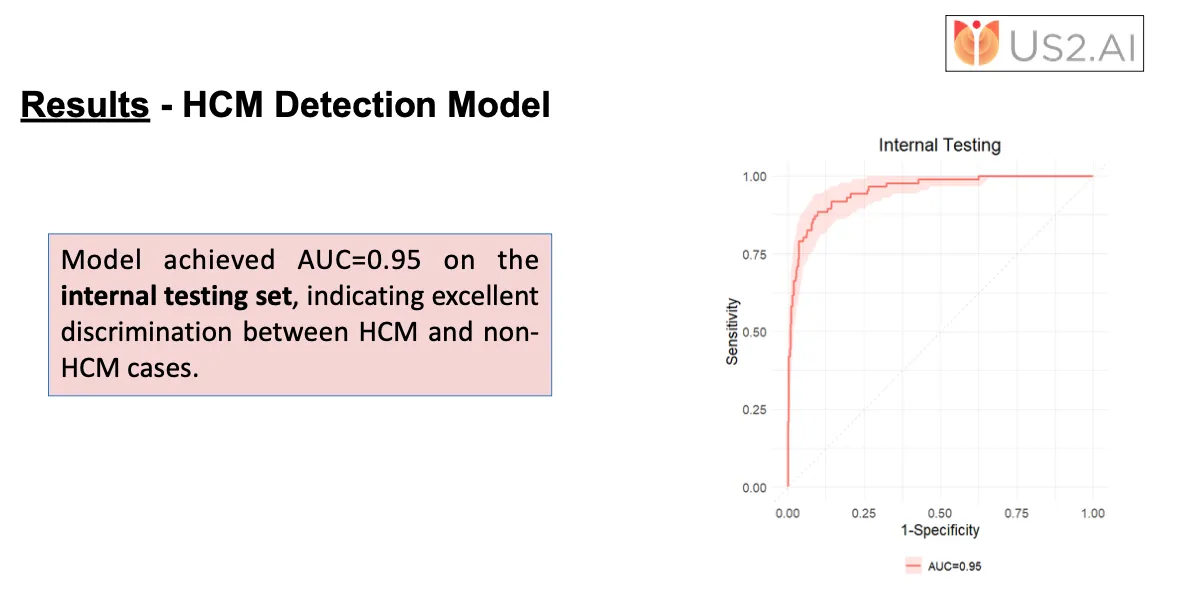
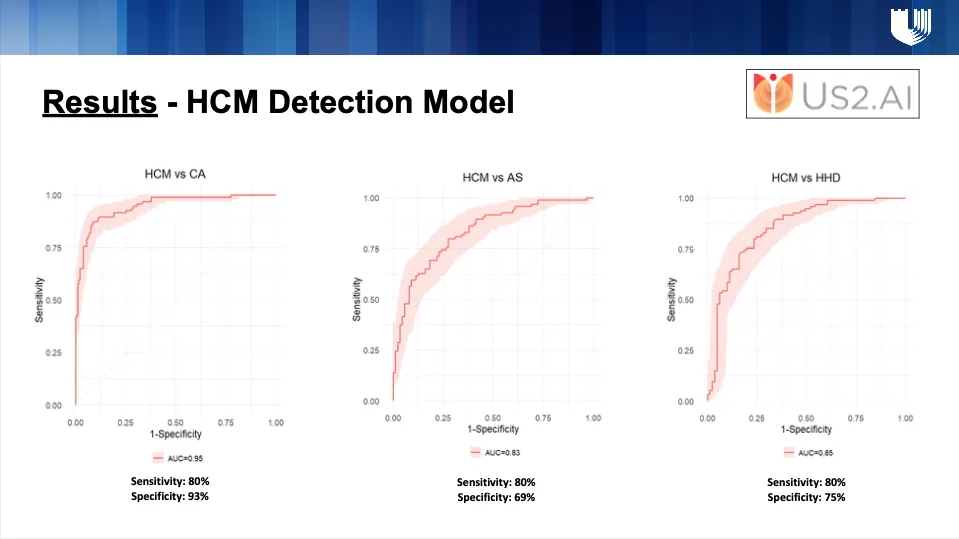
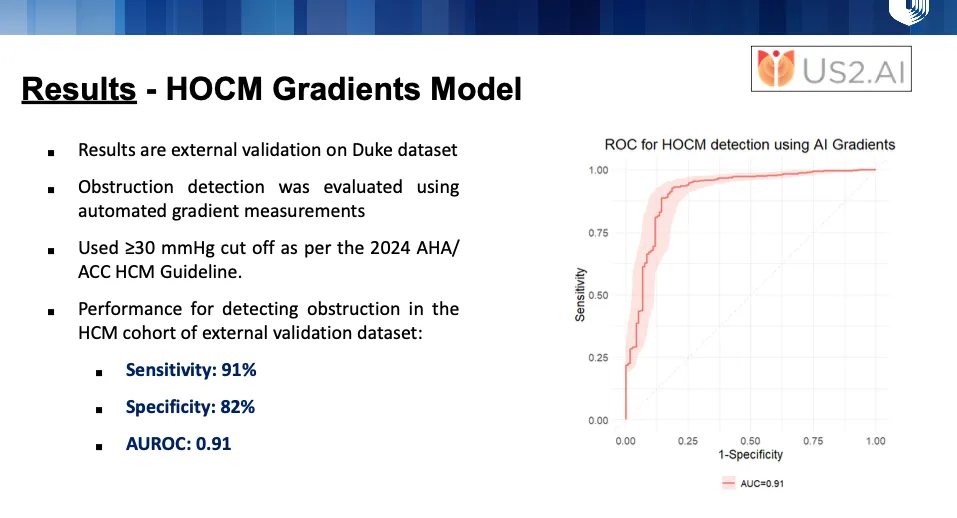
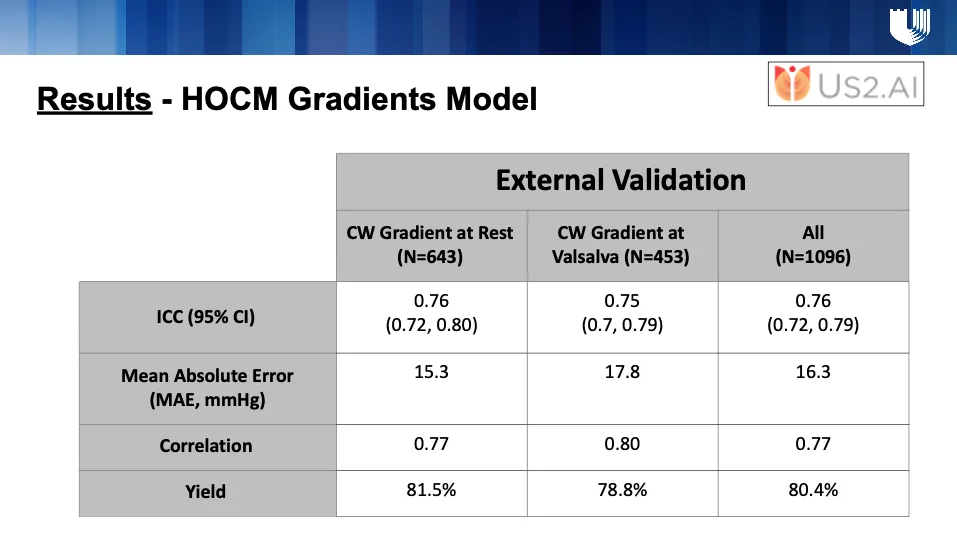
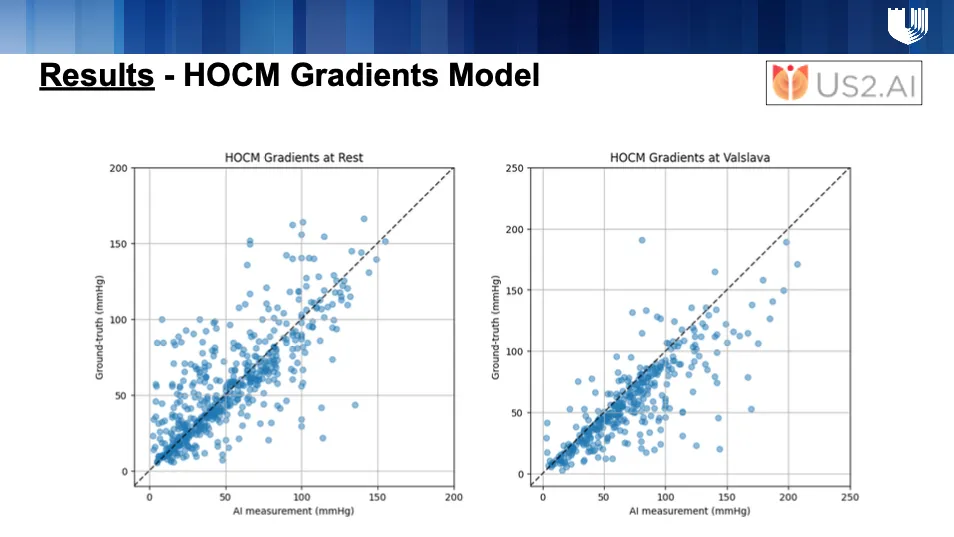

With the precision and accuracy of this model, what are some of the clinical applications of this do you see going forward in supporting HCM screening in the future?
First, it can serve as a triage or augmentation tool. Imagine a patient who undergoes a routine echo. If the echo is analyzed by this AI tool and HCM is detected, the system can flag the patient for referral to the appropriate specialists. That can lead to earlier diagnosis, appropriate treatment, and prevention of sudden cardiac death.
Second, our results show that the AI is comparable to expert cardiologists, which is remarkable.
Looking further ahead, what do you see as the future of applying AI to HCM diagnosis?
There are several exciting directions:
Multimodality integration. We envision combining this with ECG recordings, genetic information, and cardiac MRI to create a comprehensive AI tool for diagnosis and risk prediction.
Disease monitoring. This tool could be used to track disease progression and therapy response, making it valuable for longitudinal follow-up and guiding patient management.
Finally, what is one key takeaway for our audience today?
I’m very optimistic about the future. In the next 5 to 10 years, I believe every echo lab will have access to highly accurate, FDA-approved AI tools like Us2.ai. These tools will democratize expertise, allowing even rural and underserved areas without specialized cardiologists to benefit from accurate screening. This will ensure patients are diagnosed earlier and given the right treatment, reducing the risk of missed diagnoses and improving outcomes.
Media coverage by Conexiant Cardiology →