AI Echocardiography Tool Now Approved to Help Doctors Detect Cardiac Amyloidosis — A Commonly Missed Heart Disease
Singapore – July 7, 2025 – Us2.ai, a global leader in artificial intelligence (AI) for automated analysis of heart ultrasound scans (echocardiography), has announced that its latest AI Echo Copilot ™ software now includes a feature to assist in the detection of cardiac amyloidosis—a serious and frequently under-diagnosed condition. This new capability has received FDA clearance in the United States and CE mark in Europe, officially authorizing its clinical use in hospitals and healthcare settings worldwide.
Understanding Cardiac Amyloidosis
Cardiac amyloidosis is a heart condition caused by abnormal proteins building up in the heart muscle. This makes the heart stiff and less able to pump blood properly, leading to symptoms like tiredness, swelling in the legs, and shortness of breath. Hundreds of thousands of people worldwide are affected by amyloidosis, a condition that can worsen over time and may become life-threatening if left untreated.
Despite its severity, cardiac amyloidosis is commonly under-diagnosed. Research indicates that:
- Up to 1 in 4 people over the age of 85 may show signs of cardiac amyloidosis [1], and
- 13% of patients over 60 hospitalized with a certain type of heart failure may actually have undiagnosed cardiac amyloidosis [2].
Early detection of the condition is crucial, and can help ensure patients receive timely and appropriate care—especially as new treatments become available.
The Role of Echocardiography
Echocardiography, or ultrasound of the heart,is a widely used, safe, and non-invasive test to assess patients with symptoms such as breathlessness or fatigue. However, the visual signs of cardiac amyloidosis on an echocardiogram can be subtle and are often missed, particularly by less experienced readers.
How Us2.ai Enhances Detection
To address this challenge, Us2.ai’s software now includes automated detection of cardiac amyloidosis that incorporates two complementary approaches which are both FDA-cleared and CE marked:
- AI-based pattern recognition, which can detect cardiac amyloidosis from a single standard ultrasound view (the apical four-chamber, or A4C). This method has been validated in multi-center clinical studies and developed with datasets nearly twice the size of existing models, including look-alike conditions that are often mistaken as cardiac amyloidosis [3].
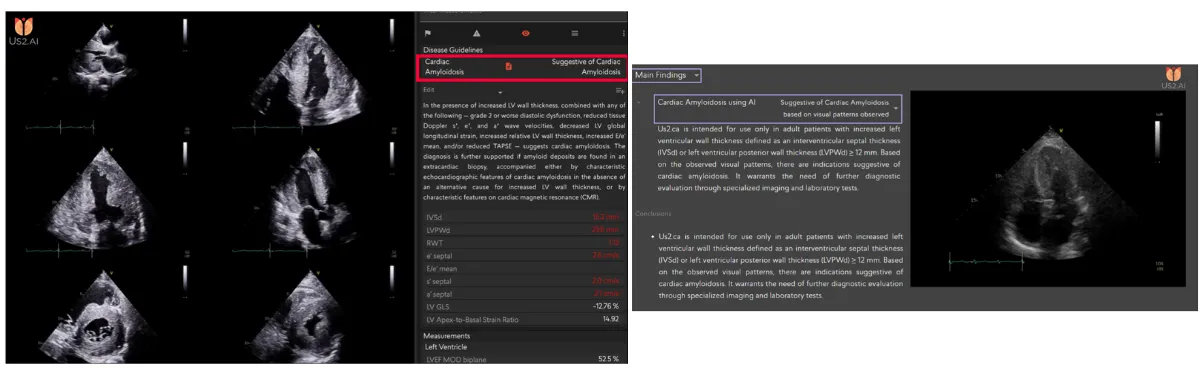
- Fully explainable automated reporting based on international clinical guidelines, using echocardiographic measurements to support standardized, evidence-based diagnosis [4-5].
Us2.ai is the first and only platform to combine both approaches — AI pattern recognition and guideline-based measurement reporting into a single software solution – providing clinicians with greater confidence and flexibility in identifying a condition that is often missed.
Beyond enhancing the accuracy of cardiac amyloidosis detection, Us2.ai’s software also plays a critical role in improving patient triage. Currently, a significant number of patients are referred to specialist centers based on test results that may incorrectly indicate the presence of cardiac amyloidosis, placing unnecessary strain on limited resources and increasing wait times for those who require urgent attention. By enabling more precise risk stratification at the point of care, whether in a referral clinic or within the specialist center, Us2.ai helps ensure that patients are appropriately directed to the right follow-up and treatment pathways. This targeted approach reduces avoidable referrals and optimizes the use of specialist time and expertise.
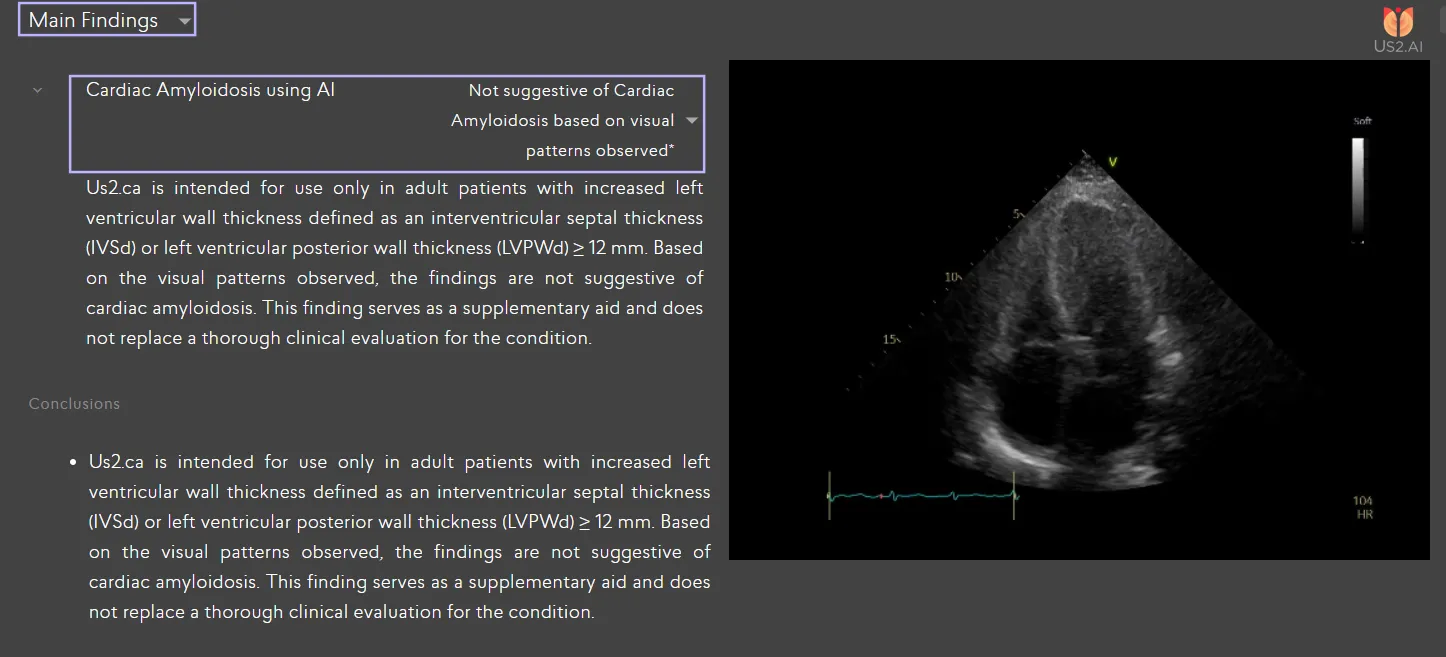
Cardiac Amyloidosis Reporting in Us2.ai
Fully explainable automated reporting based on international clinical guidelines
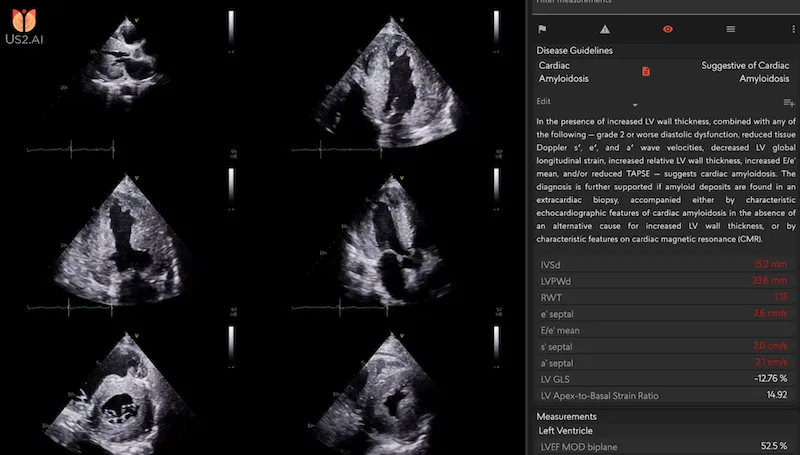
AI-based pattern recognition
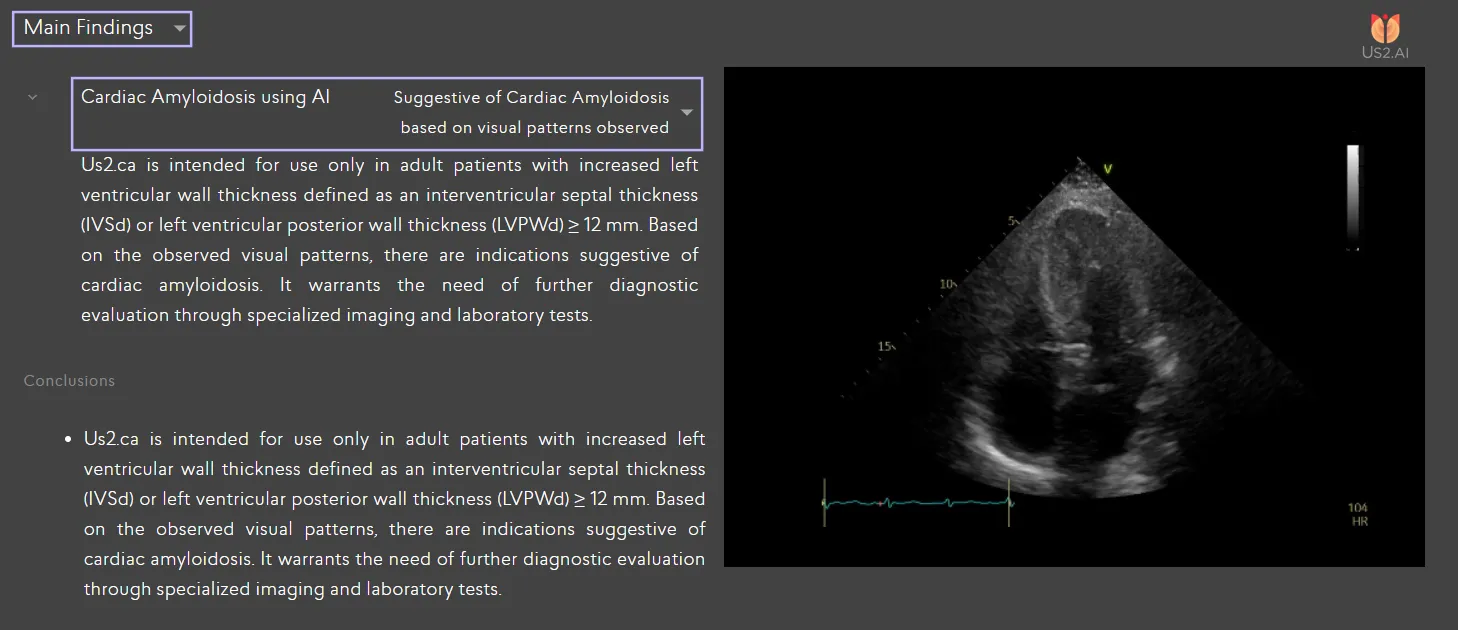
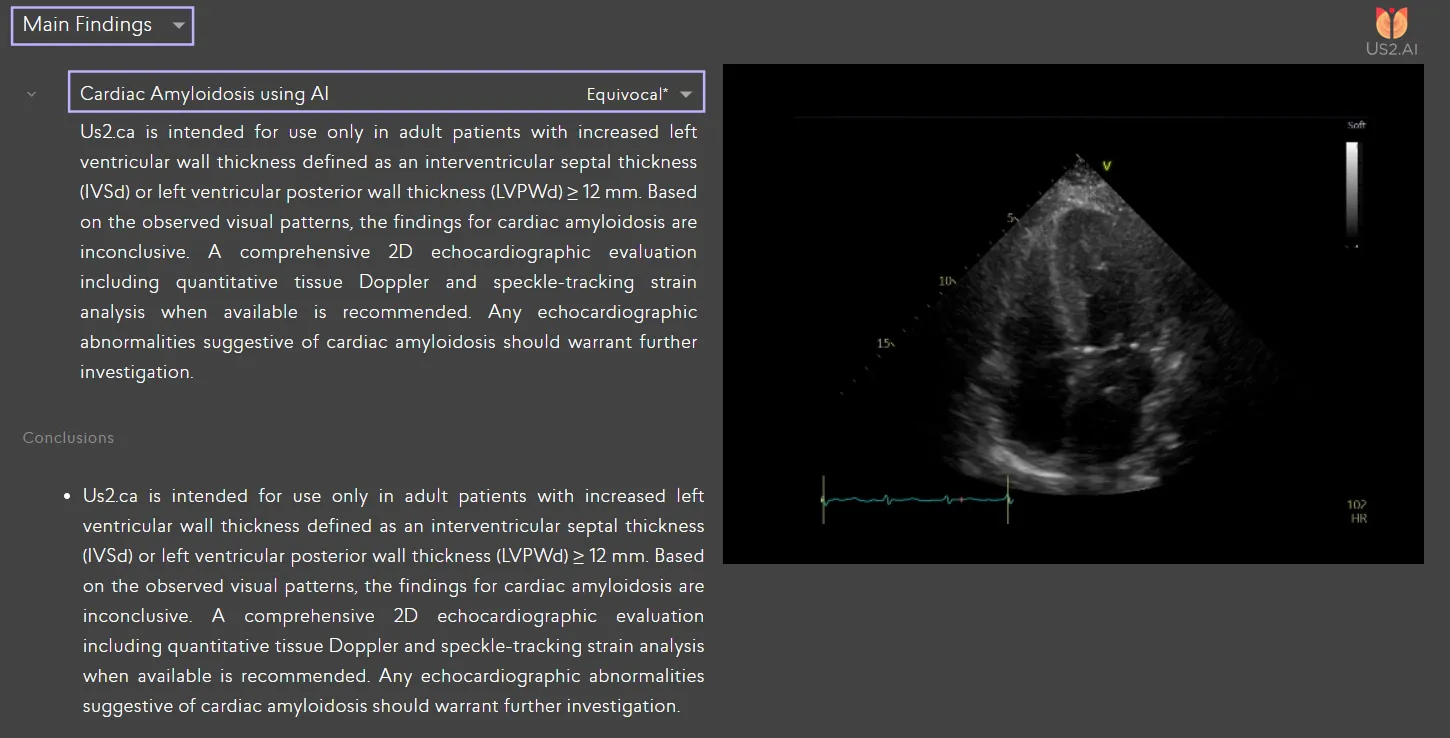
Expert Perspectives
"Cardiac amyloidosis remains a frequently under-diagnosed condition with significant prognostic implications. Us2.ai’s AI-based pattern recognition model, applied to a standard apical four-chamber view, provides a validated and scalable solution to support early detection and enhance clinical decision-making within routine echocardiographic practice.” — Marianna Fontana Professor of Cardiology and Honorary Consultant Cardiologist, National Amyloidosis Centre, University College London
"Us2.ai’s platform already provides powerful automated measurements that support the detection of cardiac amyloidosis. But this new deep learning approach moves beyond standard measurement workflows by enabling fast, fully automated detection of amyloidosis from a single echo view. By simplifying image requirements while maintaining diagnostic accuracy, it has the potential to expand the role of echocardiography as an accessible, practical screening tool for earlier identification of patients with this life threatening yet treatable disease.” – Jeremy Slivnick Assistant Professor, Division of Cardiovascular Medicine, The University of Chicago Medicine
"By embedding specialist-level AI directly into every echocardiogram—both through pattern recognition and expert measurement criteria – we hope to transform early detection of cardiac amyloidosis from a stroke of luck into standard of care—so no patient is left unseen, and no therapy arrives too late.” – Dr. Carolyn Lam Senior Consultant Cardiologist, National Heart Centre Singapore; Professor, Duke-NUS Medical School; Co-Founder, Us2.ai
Why It Matters—for Clinicians, Health Systems, and AI Leaders
The inclusion of cardiac amyloidosis detection in an AI echocardiography software marks a meaningful advancement in how artificial intelligence can support the improvement of patient care:
- For clinicians: it enhances reproducibility, efficiency, and diagnostic accuracy, enabling earlier detection and reducing the risk of missed or delayed diagnosis.
- For health systems: it provides a scalable, automated solution that aligns with international clinical guidelines – improving consistency and standardization while saving time and resources.
- For patients: it increases the chance of timely diagnosis, which can lead to earlier intervention, improved quality of life, and better long-term outcomes.
For clinical demos, implementation support, or partnership inquiries, please contact [email protected].
About Us2.ai
Us2.ai is transforming echocardiography through artificial intelligence. Our mission is to make high-quality, guideline-based heart disease diagnosis accessible to everyone—everywhere. The platform is vendor-neutral, fully automated, and backed by peer-reviewed evidence across continents.
References
[1] Tanskanen, M., Peuralinna, T., Polvikoski, T., Notkola, I. L., Sulkava, R., Hardy, J., Singleton, A., Kiuru-Enari, S., Paetau, A., Tienari, P. J., & Myllykangas, L. (2008). Senile systemic amyloidosis affects 25% of the very aged and associates with genetic variation in alpha2-macroglobulin and tau: a population-based autopsy study. Annals of medicine, 40(3), 232–239. https://doi.org/10.1080/07853890701842988
[2] González-López, E., Gallego-Delgado, M., Guzzo-Merello, G., de Haro-Del Moral, F. J., Cobo-Marcos, M., Robles, C., Bornstein, B., Salas, C., Lara-Pezzi, E., Alonso-Pulpon, L., & Garcia-Pavia, P. (2015). Wild-type transthyretin amyloidosis as a cause of heart failure with preserved ejection fraction. European heart journal, 36(38), 2585–2594. https://doi.org/10.1093/eurheartj/ehv338
[3] Ioannou A, et al. Presented at EuroEcho-Imaging 2024; 11–13 December 2024; Berlin, Germany. https://us2.ai/news/ai-echo-to-diagnose-cardiac-amyloidosis-a-multi-centre-international-development-and-validation-stu/
[4] Cotella, J. I., Randazzo, M., Maurer, M. S., Helmke, S., Scherrer‐Crosbie, M., Soltani, M., Goyal, A., Zaręba, K. M., Cheng, R. K., Kirkpatrick, J. N., Yogeswaran, V., Kitano, T., Takeuchi, M., Fernandes, F., Hotta, V. T., Vieira, M. L. C., Elissamburu, P., Ronderos, R., Prado, A., . . . Lang, R. M. (2024). Limitations of apical sparing pattern in cardiac amyloidosis: a multicentre echocardiographic study. European Heart Journal. Cardiovascular Imaging. https://doi.org/10.1093/ehjci/jeae021
[5] Ioannou, A., Patel, R. K., Razvi, Y., Hanger, M., Martinez‐Naharro, A., Venneri, L., Lim, H. S., Yoran, H., Frost, M. W., Lam, C. S., Gillmore, J. D., & Fontana, M. (2023). Automated analysis of echocardiograms at diagnosis is able to predict prognosis in ATTR cardiomyopathy. European Heart Journal. Cardiovascular Imaging, 24(Supplement_1). https://doi.org/10.1093/ehjci/jead119.387